Laura Marquez-Cantudo

| Computer-aided drug design and evaluation of enzyme inhibitors, PROTACs and LYTACs targeting cancer and other diseases |
| Laura Márquez-Cantudo1 1Departamento de Química y Bioquímica, Facultad de Farmacia, Universidad San Pablo-CEU, CEU Universities, 28668, Spain. |
| Abstract This presentation summarizes the PhD research that includes the design and computational evaluation of enzyme inhibitors (traditional and dual inhibitors), PROTACs (PROteolysis TArgeting Chimeras), and LYTACs (LYsosome TArgeting Chimeras), as new therapeutic strategies for the treatment of cancer, osteoarthritis, and amebiasis. The work combines classical and advanced tools in computational chemistry and molecular modeling.1 Osteoarthritis is a degenerative joint condition that is characterized by pain, swelling, and stiffness, thereby abruptly impacting the quality of life of mainly elderly people. Future projections to 2060 indicate that around 765 million individuals worldwide will suffer from this life-impairing pathology. Our approach relies on inhibiting Matrix Metalloproteinase 13 (MMP-13) as this enzyme is known to be overexpressed in this disease and contributes to joint degeneration by degrading collagen type II. For this, molecular docking and ADMET property prediction strategies were used to design a new promising series of inhibitors and to evaluate their druggability.2 Acanthamoeba castellanii is a free-living amoeba capable of causing keratitis in healthy individuals and fatal granulomatous encephalitis in immunocompromised patients. During its life cycle, the organism can transition from the trophozoite (infective form) to the cyst (resistant form), the latter being particularly difficult to eradicate. A leucine aminopeptidase belonging to the M17 superfamily (AcLAP) has been identified as crucial in the encystment process. In this context, inhibition of AcLAP is proposed as a promising therapeutic strategy against this amoeba. For that, four compounds designed and synthesized in our research group were repurposed and evaluated as potential inhibitors using molecular dynamics simulations. Additionally, an alternative catalytic mechanism for this enzyme is proposed based on hybrid QM/MM methods (Presented at PE Meeting 2024, OC11) Cancer is a multifactorial and highly complex pathology that is expected to affect more than 2 million people in 2026. Three different strategies were explored in this work as possible cancer treatments: a) dual inhibition of Protein Kinase 2 (CK2) and Histone Deacetylases (HDACs); b) CK2-targeting PROTACs, and c) MMP-2-directed LYTACs.3 The deregulation of these three enzymes has been associated with the proliferation, migration, and survival of tumour cells, making them attractive therapeutic targets for this disease. The binding mode of new series of these compounds was assessed by means of molecular docking and molecular dynamics simulations. In addition, to predict the permeability of the CK2-directed PROTACs, two different approaches were followed: conformational search using a Monte Carlo-combined algorithm and umbrella sampling simulations to assess their passive diffusion mechanism through plasmatic membranes (Presented at PE Meeting 2025, OC7). 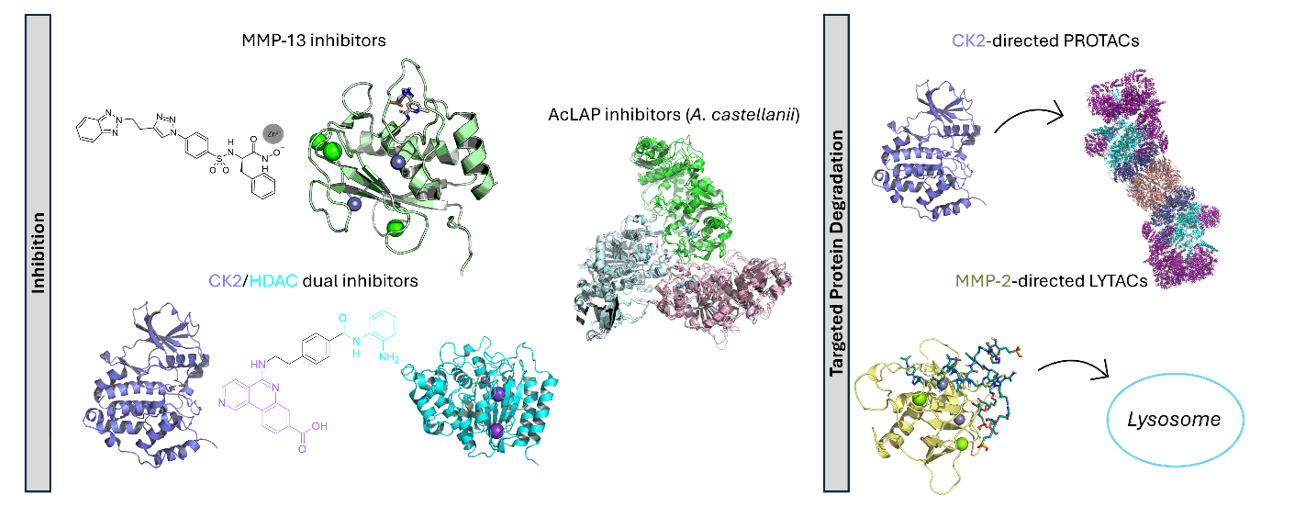 Figure 1. |
| References [1] Marquez-Cantudo L. (2025). Computer-aided drug design and evaluation of enzyme inhibitors, PROTACs and LYTACs targeting cancer and other diseases (Unpublished doctoral dissertation). Universidad San Pablo CEU. [2] Zapico J. M., Acosta L., Pastor M. et al. Int. J. Mol. Sci., 2021, 22(18), 9976. [3] Marquez-Cantudo L., Lopez-Gallego C. V., Coderch C. et al. Anales de la Real Academia Nacional de Farmacia, 2023, 89 (3), 265-285. Acknowledgments: Acknowledgements: Financial support from PID2021-123786OB-I00 (MICIU/FEDER, UE) is kindly acknowledged. L. M.-C. thanks Universidad San Pablo-CEU and Banco Santander for a Young Researcher contract. |