Sofia Vernile

FPC9 – Sofia Vernile
Sapienza University of Rome, Italy
sofia.vernile@uniroma1.it
| Benzothiophene-based monoamine oxidase inhibitors: from design to biological evaluation |
| Sofia Vernile1, Francesca Arrighi1, Michele Coluccia1, Arianna Granese1, Paola Chimenti1, Daniela Secci1, Paolo Guglielmi1 1 Dept of Chemistry and Technologies of Drugs, Sapienza University of Rome, Rome, Italy; |
| Abstract Human monoamine oxidases (hMAOs) are mitochondrial flavoenzymes that break down amines and regulate neurotransmitters such as serotonin, dopamine, and norepinephrine. Two isoforms have been identified in humans, namely hMAO-A and hMAO-B, which differ in their activity, tissue distribution and substrate specificity. The selective inhibition of the B isoform represents a kay strategy in the management of neurodegenerative disorders, including Parkinson’s disease1. Previous studies showed that benzo[b]thiophen-3-ol derivatives act as selective hMAO-B inhibitors, exhibiting neuroprotective profile in ex-vivo models2. With the aim of investigating the potential of this scaffold, two series of benzo[b]thiophen-3-ol derivatives, benzylated (Library 1, Figure 1) and acetylated (Library 2, Figure 1), were designed and evaluated for their inhibitory activity in this study. Acetylated compounds were generally strong hMAO inhibitors but lacked selectivity, affecting both hMAO-A and hMAO-B. In contrast, benzylated compounds showed activity and selectivity depending on the type and position of substituents on the aromatic ring. These findings highlight the critical role of structural modifications in modulating isoform selectivity and support the further optimization of benzylated derivatives as promising hMAOs inhibitors. 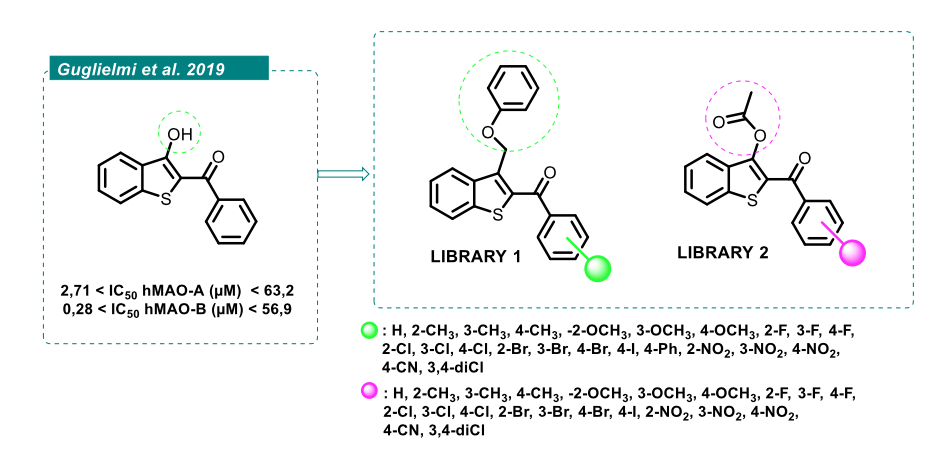 Figure 1. Design of the novel derivates. |
| References [1] Edmondson, D. E.; Binda, C. Subcellular Biochemistry; Springer Singapore: Singapore, Vol. 87 (2018) [2] Guglielmi, P. et al. Benzo[b]thiophen-3-ol derivatives as effective inhibitors of human monoamine oxidase: design, synthesis, and biological activity. J Enzyme Inhib Med Chem 34, 1511–1525 (2019) |