Yushun Ihor

| Synthesis and biological activity of thiazolidine derivatives with a pyrazoline fragment in molecules |
| Yushyn Ihor1 1 Department of Pharmaceutical, Organic and Bioorganic Chemistry, Danylo Halytsky Lviv National Medical University, 69 Pekarska St., 79010 Lviv, Ukraine |
| Abstract Pyrazoline–thiazolidine hybrids are considered promising scaffolds because of their ability to interact with multiple biologically relevant targets, including kinases, inflammatory mediators, and nuclear receptors, which determines their broad pharmacological potential [1,2]. The dissertation focused on the synthesis of novel pyrazoline-containing thiazolidine and related derivatives as promising bioactive heterocycles. Efficient synthetic approaches, including [2+3]-cyclocondensation, Knoevenagel condensation, Claisen–Schmidt reaction, and hetero-Diels–Alder reaction, enabled the preparation of structurally diverse pyrazolyl-substituted 4-thiazolidinones, thiazoles, and fused heterocyclic systems. Pyrazole-derived carbothioamides proved to be effective S,N-binucleophilic reagents for obtaining new thiazolidinone derivatives, while reactions of isorhodanine with 3-methyl-1H-pyrazol-5-amine afforded key intermediates for further functionalization. Unexpected transformations led to the formation of condensed pyrazolo[3,4-b]thiazolo[5,4-e]pyridines. Structures were confirmed by LC–MS, NMR, and X-ray analysis. Biological screening identified compounds with antitumor, antimicrobial, antifungal, and anti-inflammatory activities; some active derivatives also suggested possible PARP1/2 inhibition. The most active compounds showed selective cytotoxicity toward Jurkat and MCF-7 cell lines, while selected fused heterocycles demonstrated activity against Gram-positive microorganisms. Overall, 70 compounds were synthesized, and 20 hit compounds were identified. 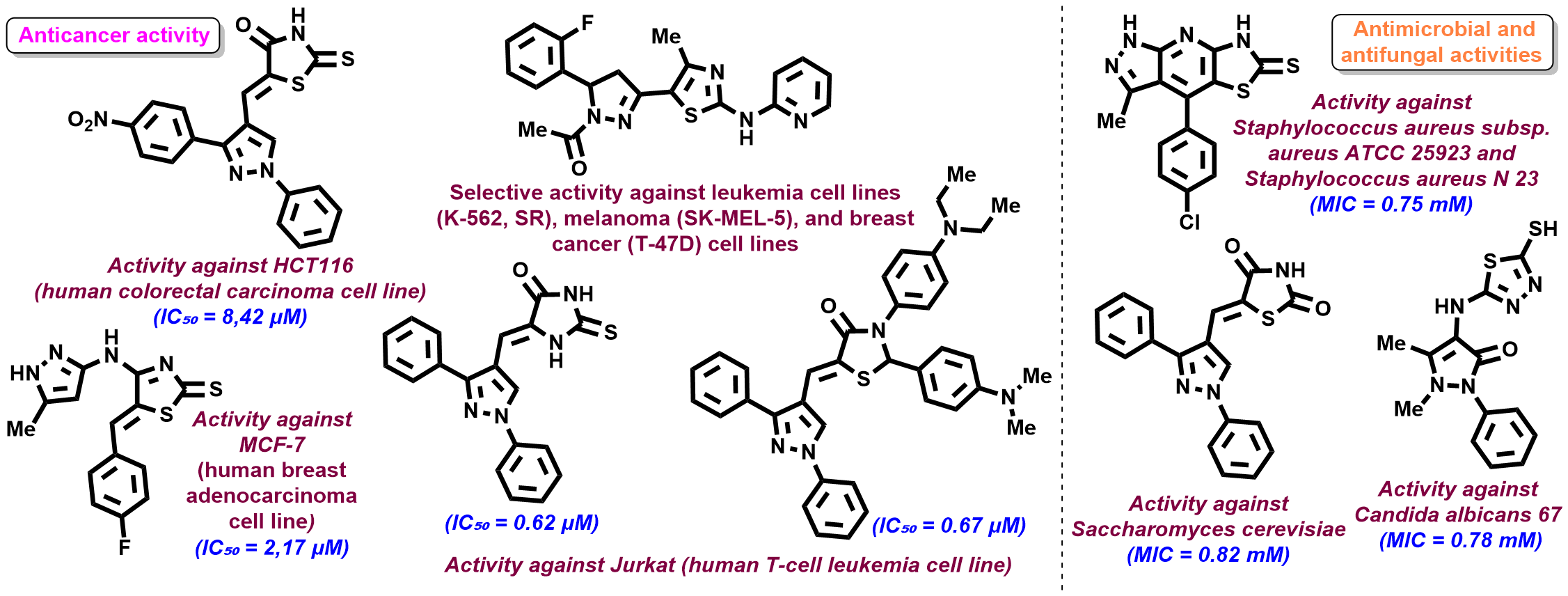 Figure 1. The most active synthesized molecules |
| References [1] Havrylyuk, D.; Roman, O.; Lesyk, R. Synthetic approaches, structure activity relationship and biological applications for pharmacologically attractive pyrazole/pyrazoline–thiazolidine-based hybrids. Eur. J. Med. Chem. 2016, 113, 145–166. https://doi.org/10.1016/j.ejmech.2016.02.030 [2] Elewa, S.I.; El-Farargy, A.F.; Nafie, M.S.; Mansour, E. Synthesis, and Cytotoxic Activity of Novel Pyrazoline-Thiazolidinone Derivatives with Molecular Docking Studies. Polycycl. Aromat. Compd. 2023, 43, 5807–5825. https://doi.org/10.1080/10406638.2022.2108074 |