Manuel Novás

OC13 – Manuel Novás
1. BioFarma Research Group, Center for Research in Molecular Medicine and Chronic Diseases (CiMUS), Universidade de Santiago de Compostela, 15782 Santiago de Compostela, Spain
2. Department of Organic Chemistry, Faculty of Pharmacy, University of Santiago de Compostela, 15782, Santiago de Compostela, Spain
| Pharmacomodulation of 2-Amidobenzothiazoles for the Development of Neuroprotective Compounds: A Phenotypic Study in an Alzheimer’s Disease Model |
| Novás Manuel1,2, Martínez Antón L. 2, Loza María Isabel 2, and Matos Maria J. 2 1 BioFarma Research Group, Center for Research in Molecular Medicine and Chronic Diseases (CiMUS), Universidade de Santiago de Compostela, 15782 Santiago de Compostela, Spain; 2 Department of Organic Chemistry, Faculty of Pharmacy, University of Santiago de Compostela, 15782, Santiago de Compostela, Spain; |
| Abstract The benzothiazole scaffold and its analogs are present in several clinically approved central nervous system drugs with neuroprotective properties, including riluzole, pramipexole (and dexpramipexole), and lurasidone. Inspired by the therapeutic potential of this privileged structure, we designed and synthesized a library of 48 novel 2-amidobenzothiazole derivatives through systematic pharmacomodulation of the scaffold. Structural diversity was introduced by incorporating a broad range of electron-donating and electron-withdrawing groups, as well as substituents of varying steric demand, in order to expand the explored chemical space. Compounds were then screened in a phenotypic Alzheimer’s disease model of SH-SY5Y cells transfected with the mutations MAPT P301L and APP V717I, which induce the formation of pathological protein aggregates [1]. A high-throughput screening calcium influx assay and neurite length immunostaining were performed to determine the neuroprotective effect of the compounds. Screening of the compound library identified derivatives capable of producing a dose-dependent protective effect, reversing neurite shortening and attenuating calcium influx alterations in the cellular model. Physicochemical profiling revealed that trifluoromethyl and trifluoromethoxy substituents significantly modulate compound solubility, which in turn influences the observed biological activity [2]. The study of this family of 48 new compounds has helped establish interesting structure-activity relationships, which may enable further optimization of the scaffold in the hit-to-lead process. 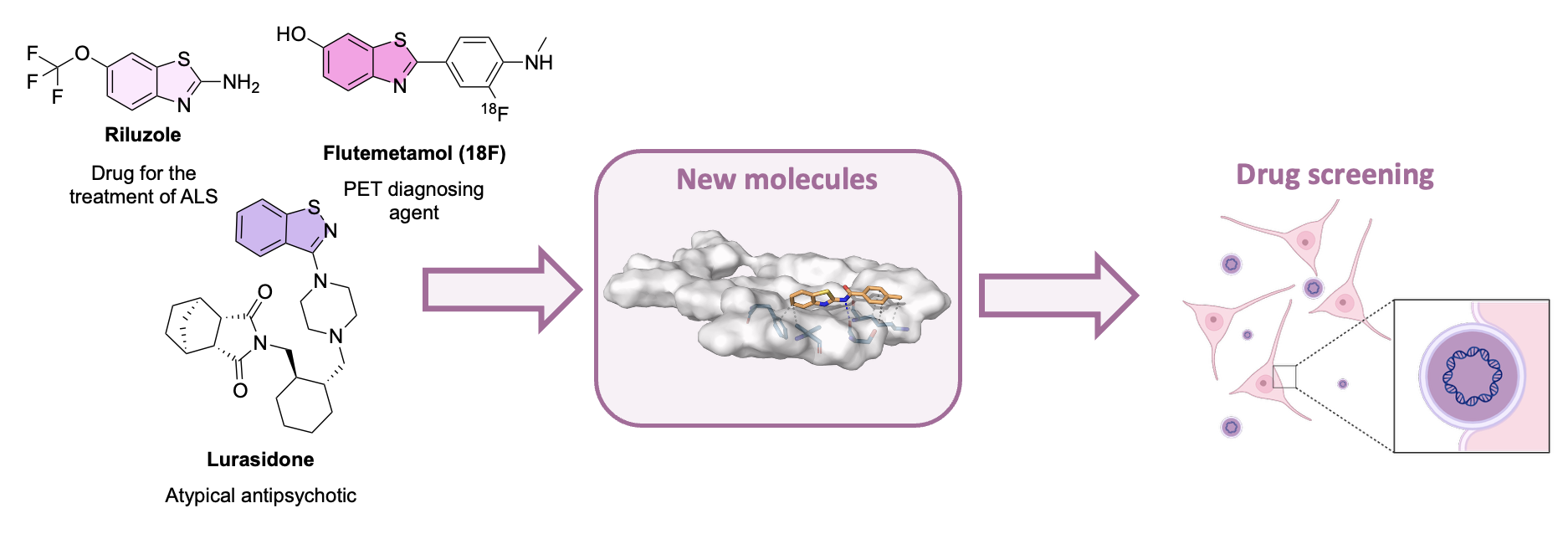 Figure 1. Diagram of the workflow of the project. |
| References [1] Barro-Fernández, M. Development of a novel in vitro model of Alzheimer’s disease. PhD thesis, Universidad de Santiago de Compostela, 2025. [2] Novás, M; Matos, M.J. The Role of Trifluoromethyl and Trifluoromethoxy Groups in Medicinal Chemistry: Implications for Drug Design. Molecules 2025, 30, 3009. https://doi.org/10.3390/molecules30143009. |