Leonardo Lemos

OC6 – Leonardo Lemos
Department of Organic Chemistry, Faculty of Pharmacy, University of Santiago de Compostela, 15782, Santiago de Compostela, Spain.
leonardo.censi@rai.usc.es
| Synthesis, characterization and evaluation of anti-T. cruzi activity of 1-arylpyrazole-4-carboximade-N-propylimidazoles |
| Lemos Leonardo C1,2, Santos Maurício S. 1 and Matos Maria J. 2 1 Department of Physic and Chemistry, Federal University of Itajubá, Itajubá, 37500-903, Brazil. 2 Department of Organic Chemistry, Faculty of Pharmacy, University of Santiago de Compostela, 15782, Santiago de Compostela, Spain. |
| Abstract Chagas Disease (CD), an illness first identified by Carlos Chagas 117 years ago, continues to represent a significant public health challenge. An annual total of thirty thousand new cases has been reported, resulting in fourteen thousand deaths during the same period. At the moment, only two drugs are used in the chemotherapy: benznidazole and nifurtimox. Nifurtimox is the only drug commercially available in several countries. The primary concerns regarding this drug are due to its high rate of attrition, which is estimated to be approximately 20%, largely attributable to the occurrence of substantial adverse effects. Additionally, the prolonged duration of its administration is another important issue, as is its apparent inefficacy in the chronic phase of CD [1]. In the search for new candidates with potential trypanocidal effect, our research group has been synthesizing several heterocyclic systems for their antiparasitic evaluation [2]. The main goal of this study is to synthesize and evaluate the trypanocidal activity of 13 novel 1-aryl-N-(3-(1H-imidazol-1-yl)propyl)-1H-pyrazole-4-carboxamides. The structures of all the compounds have been confirmed by Fourier-transform infrared spectroscopy, high-resolution mass spectrometry and nuclear magnetic resonance analysis. Biological evaluation was conducted on the clone Dm28c in both its intracellular amastigote and trypomastigote forms. This work will present the initial chemical and biological results. 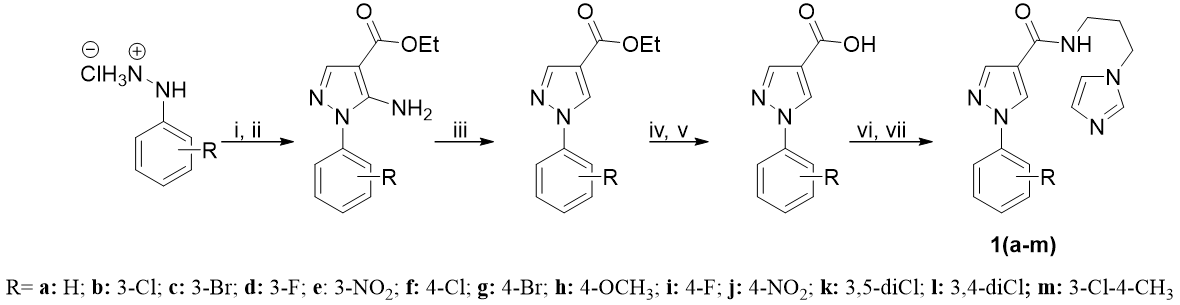 Scheme 1. Synthetic route to obtain 1(a-m). Reagents and conditions: i) AcONa, ethanol, 15 min; ii) ethyl-2-cyano-3-ethoxyacrylate, 1 h, reflux; iii) t-butyl nitrite, THF, 2 h, reflux; iv) NaOH 3,5 mol*L-1, ethanol, 10 min, reflux; v) HCl 10% m/v; vi) SOCl2, toluene, 70 ◦C, 2 h; vii) 3-amine-1-(1H-imidazol-1-yl)propane, pyridine, 70 ◦C, 2 h. |
| References [1] Durães-Oliveira, J.; Palma-Marques, J.; Moreno, C.; Rodrigues, A.; Monteiro, M.; Alexandre-Pires, G.; Fonseca, I. P.; Santos-Gomes, G. Chagas Disease: A Silent Threat for Dogs and Humans. Int. J. Mol. Sci. 2024, 25, 3840-3865. https://doi.org/10.3390/ijms25073840. [2] Orlando, L. M. R.; Lechuga, G. C.; Lara, L. S.; Ferreira, B. S.; Pereira, C. N.; Silva, R. C.; Santos. M, S.; Pereira, M. C. S. Structural Optimization and Biological Activity of Pyrazole Derivatives: Virtual Computational Analysis, Recovery Assay and 3D Culture Model as Potential Predictive Tools of Effectiveness against Trypanosoma cruzi. Molecules 2021, 26, 6742-6770. https://doi.org/10.3390/molecules26216742. |