Ewout Van de Velde
| Generalized parallel synthesis and anti-trypanosomal evaluation of a chemically diverse 6-functionalized C-nucleoside compound library |
| Ewout Van de Velde1, Lisa Marriottini1, Anouk Van Hauwermeiren1, Guy Caljon2, Maria de Nazare Correira Soeiro3, Serge Van Calenbergh1 1 Ghent University, Faculty of Pharmaceutical Sciences, Laboratory for Medicinal Chemistry. 2 University of Antwerp, Faculty of Pharmaceutical, Biomedical and Veterinary Sciences, Laboratory of Microbiology, Parasitology and Hygiene 3 Laboratório de Biologia Celular do Instituto Oswaldo Cruz, Fiocruz, Avenida Brasil 4365, Manguinhos, Rio de Janeiro, Brazil |
| Abstract Neglected Tropical Diseases (NTDs) caused by vector-borne protozoan parasites—including African sleeping sickness, Chagas disease, and leishmaniasis—disproportionately affect impoverished populations in tropical and subtropical regions. Current treatments suffer from toxicity, complex administration, and emerging resistance, underscoring the urgent need for safer, selective antiparasitic agents. C-Nucleosides, defined by a carbon–carbon glycosidic bond, resist enzymatic cleavage by hydrolases and phosphorylases and allow systematic variation in ring composition to generate novel pharmacophores. The demonstrated in vivo activity of 9-deazainosine and formycin B against Trypanosoma and Leishmania highlights their therapeutic potential, yet efficient access to structurally diverse C-nucleoside libraries remains limited. Here we report a late-stage diversification platform, enabled by a 3,5-bis(trifluoromethyl)phenoxy (BTFPO) handle, that allows rapid assembly of C-nucleosides bearing three distinct purine-like scaffolds and a wide range of substituents from stable, versatile intermediates. This approach consistently delivers potent, selective anti-trypanosomatid agents, as demonstrated by the in vivo efficacy and safety of lead compound 58 in two distinct mouse models. 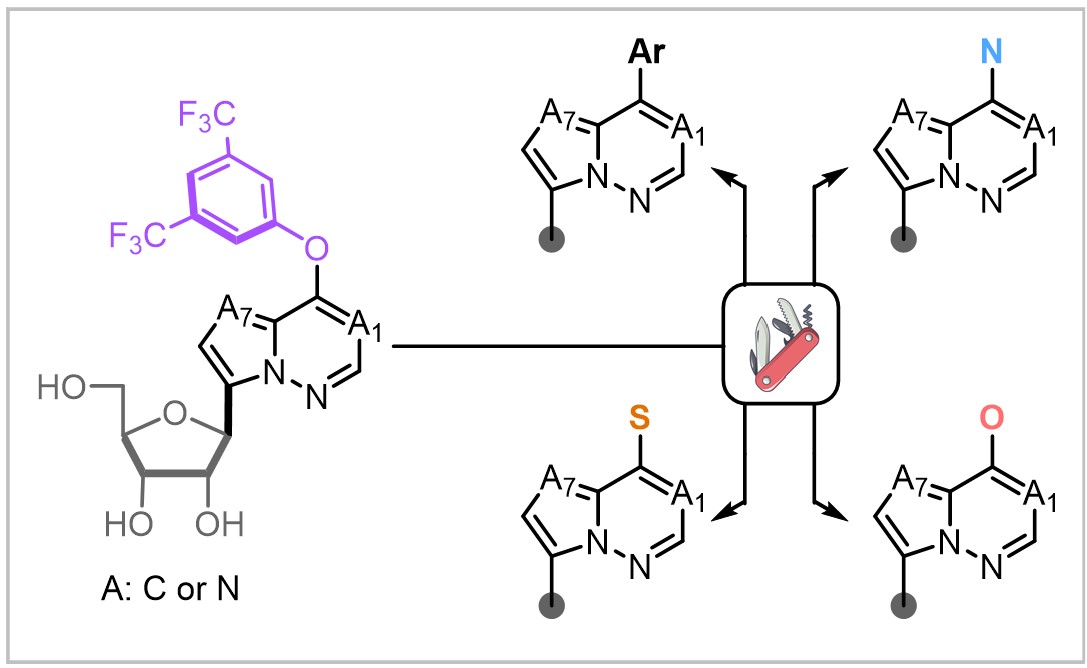 Figure 1: Late-stage functionalization of C-nucleoside analogues by use of BTFPO |
| References [1] Avila, J. L.; Polegre, M. A.; Robins, R. K. Biological Action of Pyrazolopyrimidine Derivatives against Trypanosoma Cruzi. Studies in Vitro and in Vivo. Comp. Biochem. Physiol. C 1987, 86 (1), 49–54. https://doi.org/10.1016/0742-8413(87)90143-5. [2]Berman, J. D.; Hanson, W. L.; Lovelace, J. K.; Waits, V. B.; Jackson, J. E.; Chapman, W. L. J.; Klein, R. S. Activity of Purine Analogs against Leishmania Donovani in Vivo. Antimicrob. Agents Chemother. 1987, 31 (1), 111–113. https://doi.org/10.1128/AAC.31.1.111. |
