Sara Salvi

FPC7 – Sara Salvi
Department of Drug Sciences, University of Pavia, Via Taramelli 12, 27100, Pavia (PV), Italy
sara.salvi01@universitadipavia.it
| Dimeric Sigma Receptor-Proteasome Ligands as Anticancer Agents for Glioblastoma |
| Salvi Sara1, Rossino Giacomo1, Vaccari Melania Elettra1, Malacrida Alessio2, Molteni Laura2, Miloso Mariarosaria2, Collina Simona1 1 Department of Drug Sciences, University of Pavia, Via Taramelli 12, 27100, Pavia (PV), Italy 2 Department of Medicine and Surgery, University of Milano-Bicocca, Via Cadore 48, 20900, Monza (MB), Italy |
| Abstract Glioblastoma multiforme (GBM) is one of the most aggressive and lethal brain tumors, accounting for more than half of all malignant brain tumors [1]. This work aims to identify a new generation of anticancer agents for GBM treatment through a multitarget-directed ligand (MTDL) approach, simultaneously targeting Sigma receptors (SRs) and the proteasome, whose interconnected roles support their combined modulation. In previous studies, we identified two hit compounds targeting SRs and the proteasome, respectively: RC106 and IP24 (Figure 1). Both compounds exhibit antiproliferative activity against various cancer cell lines [2,3]. In the present study, these two scaffolds were covalently combined to generate a small set of dimeric MTDLs. RC106 and IP24 were linked through different spacers to achieve dual SR-proteasome modulation and improved antitumor efficacy. A total of four dimeric compounds were synthesized, exploring the effect of different derivatization strategies and inter-pharmacophore distance. 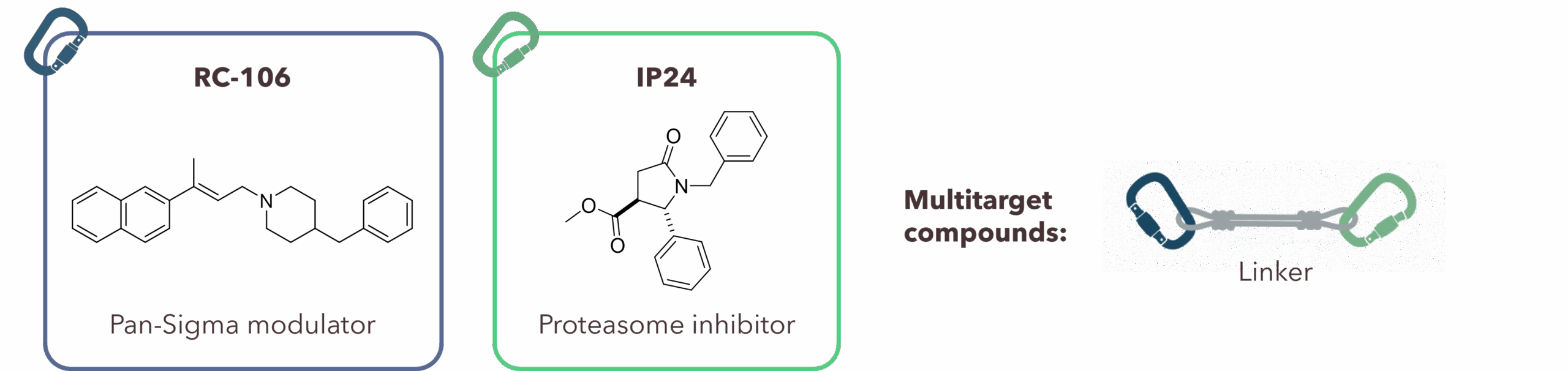 Figure 1. Chemical structure of RC106 and IP24, and schematic representation of final dimeric compounds. To assess whether the dimeric architecture could compromise target engagement, the conformation of a representative compound was investigated in aqueous solution using one- and two-dimensional NMR experiments. The experimental data, supported by computational studies, showed that the dimer adopts an extended conformation, which is crucial for effective dual target interaction. Biological evaluation is currently ongoing, and includes antiproliferative assays on GBM cell lines, as well as the assessment of SRs binding affinity and proteasome inhibition, in order to confirm the proposed multitarget mechanism of action. |
| References [1] Królikowska, K et al. Int. J. Mol. Sci. 2025, 26 (24), 12162. https://doi.org/10.3390/ijms262412162. [2] Rui, M. et al. Eur. J. Med. Chem. 2016, 124, 649–665. https://doi.org/10.1016/j.ejmech.2016.08.067 [3] Listro, R. et al; Int. J. Mol. Sci. 2022, 23 (21), 13061. https://doi.org/10.3390/ijms232113061 |