Angela Pagliaro

| Structure-Based Design of RhoA Effector Interaction Inhibitors for the Treatment of Cancer |
| Pagliaro Angela1,2, Brindani Nicoletta1, Bergamaschi Enrico1, Vitali Forconesi Gabriella1, Maria Antonietta La Serra1, Veronesi Marina1, Cardoso De Azevedo Moreira Miguel Angelo1, Bonaldo Valerio1, Girotto Stefania1, De Vivo Marco1 1 Italian Institute of Technology (IIT), Genova, Italy 2 Department of Pharmacy and Biotechnology Fabit, University of Bologna, Bologna, Italy |
| Abstract Rho GTPases are a family of small G proteins that regulate essential physiological processes, including cytoskeletal dynamics and cell cycle progression. Our research laboratory has been actively involved in the field of protein-protein Interactions. Previous studies focused on disrupting the complex between the GTPase CDC42 and its effector PAK protein, leading to the identification of lead compounds with anticancer activity in vivo. These have shown a good pharmacokinetic profile, bioavailability, and efficacy in multiple in vivo PDX tumor mouse models [1]. Building on these results, we have now turned our focus toward inhibiting the RhoA/ROCK1 interaction. This plays a pivotal role in cancer pathology, with RhoA being overexpressed or hyperactivated in several tumors. In melanoma, the activation of RhoA and its downstream kinase, ROCK1, is directly correlated with disease progression through deregulation of cell motility [2,3]. We started with a virtual screening and identified twenty candidate compounds. These underwent an intrinsic fluorescence assay to test their binding with RhoA. Three compounds were identified as promising hits. Synthesis of a few analogs confirmed their activity and a favorable DMPK profile. Based on these preliminary data, we are currently pursuing further optimization of these promising compounds to block RhoA. 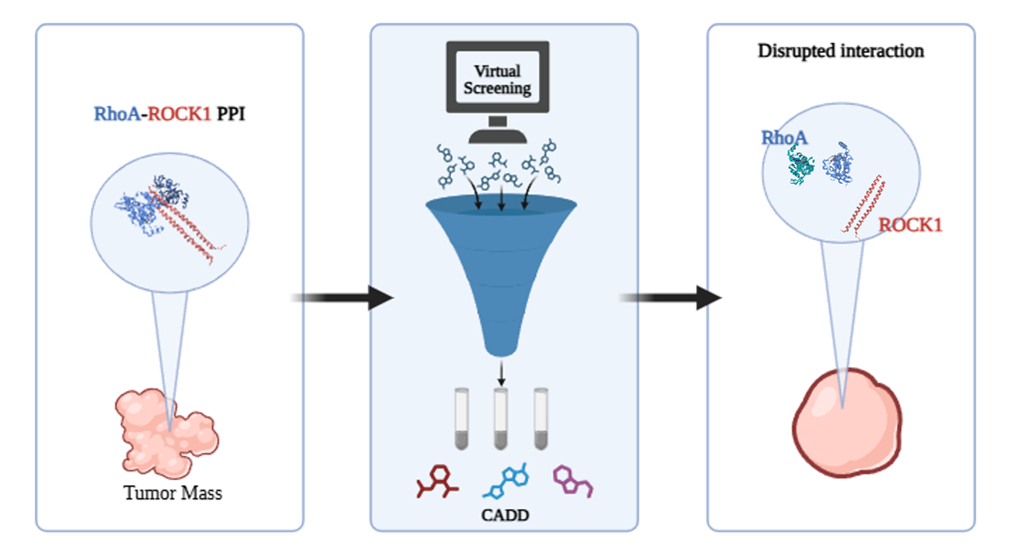 Figure 1. Project Workflow |
| References [1] Brindani, Nicoletta, et al. “Discovery of CDC42 Inhibitors with a Favorable Pharmacokinetic Profile and Anticancer In Vivo Efficacy.” Journal of Medicinal Chemistry 67.12 (2024): 10401-10424. [2] Crosas-Molist, Eva, et al. “Rho GTPase signaling in cancer progression and dissemination.” Physiological Reviews 102.1 (2022): 455-510. [3] Julian, Linda, and Michael F. Olson. “Rho-associated coiled-coil containing kinases (ROCK) structure, regulation, and functions.” Small GTPases 5.2 (2014): e29846. |